A Backbone of Research
How Suna Sibi is helping advance ASG’s focus on research and client collaboration
ASG’s own Suna Sibi, a technical project manager and business line lead, recently co-authored an article published in Expert Opinion on Drug Delivery (EODD), a peer-reviewed journal focused on drug delivery, development and research.
“We’re so proud of Suna,” said Meagan Koeneman, who leads marketing and business development at ASG. “She’s committed to taking science and her work to the next level, and this publication proves it. Curiosity and service are ASG values, and Suna is living them.”
The article, “A multimodal perspective to overcome translational barriers in cerebrospinal drug delivery,” which Suna co-authored with Christopher Kadamus, Reshma Bharadwaj and J. Anand Subramony, explores the complexities of delivering drugs directly to the central nervous system. It focuses on cerebrospinal fluid, the liquid that cushions and nourishes the brain and spinal cord. Drug therapies can be delivered directly into cerebrospinal fluid via various locations in the brain and spine, allowing these treatments to quickly reach targeted areas.
Working on the research paper gave me freedom to think and play around with different ideas.
So far, most research on cerebrospinal drug delivery has been based on animal models, Suna said. It has been a challenge to translate data from research using animal models to applications for the human brain. Suna’s study addresses this issue by underscoring the importance of translating animal models to human applications and the value of visualization during the testing of drug delivery. Suna and her collaborators had the goal of offering practical advice on how to conduct preclinical trials and translate findings from animals to human models effectively.
I recently spoke to Suna to find out more about the publication and her research process.
ABOUT THE STUDY
What’s the focus of this study?
Addressing the challenges and recommendations associated with drug delivery into the central nervous system. So anywhere in the body via cerebral spinal fluid. by your spine and brain via cerebral spinal fluid. It’s a review article focused on our discussions with expert consultants and then subsequent literature search to support our findings.
I hope people can use our work to standardize best practices for drug delivery research.
There are three major categories to the publication: First, the translation aspect between doing studies in animals and bringing that to clinical settings in humans. Then we had to take into account neuroimaging: using the appropriate type of scanner to visualize what’s happening when doing drug delivery in this fashion. Finally, you need to be able to characterize how the cerebral spinal fluid flows and what kind of delivery parameters to use to balance putting a drug into something and making sure it doesn’t destabilize what you’re injecting into.
What’s cerebrospinal fluid-mediated drug delivery?
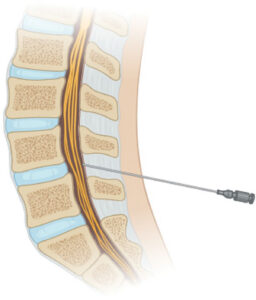
Cerebral spinal fluid is a bit like the plumbing system in the human body. It’s fluid that flows around our brain and spinal cord like water flows through a pipe, and it helps regulate the pressure in our skull. It also acts as a shock absorber for the brain and spinal cord. It can be regulated by factors like breathing, heart rate and posture. The reason delivery into cerebrospinal fluid is being researched is because it helps bypass the blood-brain barrier, which is really selective about what goes through it to reach the brain. This is commonly done in clinical practice via lumbar puncture and is mainly for anesthesia and pain management, but it’s also being evaluated for gene therapy potential. The blood-brain barrier is just a bunch of different cells packed together that selectively lets things through it from the bloodstream and into the brain. It’s designed this way so not just anything can get into your brain and infect it. Cerebrospinal fluid isn’t nearly as selective about what it takes in.
Why is it important to research cerebrospinal fluid drug delivery?
Cerebrospinal fluid-mediated drug delivery is a convenient way to get around the blood-brain barrier through a minimally invasive means of injecting into the body. Lower lumbar punctures are already commonly done in clinical practice, so expanding the practice isn’t too much of a stretch. It’s helpful and important to leverage current clinical practice in drug delivery development to minimize time to market.
PROJECT BACKGROUND
Tell us about your publishing experience.
This is actually my first publication ever! The study was an abstracted, fun way for me to investigate something before it became a fully conceptualized idea, which you’d typically see in the active commercialization space. Working on the research paper gave me freedom to think and play around with different ideas. The process has definitely been a journey. It wasn’t until a few days after the paper was published, when people started reaching out and providing words of recognition, that I began thinking: This really is a big thing that we did! I was happy to be a part of the collaboration, and I’m proud of what it turned into.
How did you develop the idea behind the article?
The project started out as a journal entry that I had put together as part of supporting a client on a project, just saying, “We consulted these experts; here’s what we learned.” My client’s boss heard about it, and he thought it had potential, so he told us to do more work with it. So we took what I had and then supported it with additional literature findings, and then came up with this comprehensive summary that became the publication you see today.
How did your team conduct the research?

We each had our own role. Chris Kadamus is the client I was working with. He has a background in device development, and he’s done a lot of deep diving into central nervous system drug delivery. He’s essentially the technical brains behind this project.

Reshma Bharadwaj is a research specialist who works at Lilly’s Bengaluru, India, site. Whenever we have a request for a comprehensive literature search or technical writing to support an investigation for a given project, we loop in her team. She’s got a lot of familiarity with what these scientific publications require, so she was a valuable resource. She made sure we met the formatting and content guidelines. She also helped make sure we had appropriate literature support for what we were claiming as expert opinions.

Anand Subramony was essentially the sponsor. He was the one who originally told us to pursue this project. He gave us a lot of really good advice on some of the common principles of publication writing and gave us pointers on finding specific journal guidelines. He was basically the organizational oversight and advisory. He was very supportive in helping see us the project through to completion.

I was the project manager. I helped coordinate all of the engagements and interactions with the experts we referenced in the article. I helped make sure we got the right folks in the right room talking about the right kind of thing and made sure we stayed on target, meeting our goals and expectations. I kept a closely documented log of the process we followed, and I also wrote a high-level description of the method in the article and the supplemental section that goes into detail about how we went about the process.
What’s the role of neuroimaging in this research?
When you’re injecting into something, especially something with such a delicate pressurized system like a human being, you want to be able to see what’s happening. That’s where neuroimaging comes into play. MRI is the most commonly used scanner, but it helps to combine it with another
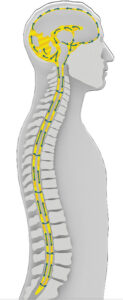
scanning technique that can actually help you track drug molecules that you can label. Just because what you’re seeing what’s happening doesn’t mean you know the thing that’s flowing is the thing you actually want to track. You need to find a way to use a scanning technique that gets you the resolution you want at the depth you want while also making sure you can see what you’re trying to see. Imaging like this also helps you get a feel for how the body system changes over time due to age versus disease.
It’s also important to characterize how cerebrospinal fluid flows under different conditions. Detailed imaging can help determine what delivery parameters you use, since the flow is influenced by heartbeat, breathing and other anatomical features. It can flow completely differently depending on whether you’re sitting, standing, on your side, etc., and that in turn will influence what kind of delivery parameters you can use, injection rate and how much volume you can use. Imaging also helps a researcher see how the drug spreads in the brain.
FROM ANIMALS TO HUMANS
What’s translational research in the context of your study?
Translational research bridges the gap between basic scientific discoveries and practical applications. Before you test in humans, you have to determine what’s safe in something else. When it comes to drug delivery inside the body, you typically start by testing in animals. In the case of this type of drug delivery system, into the central nervous system, we found that one animal model doesn’t seem to replicate how everything works. What we recommend instead is a tiered approach. You want to start with the basics before moving onto more complicated models. Animals like rats are your foundational research. You can get principles behind the dosing mechanics, and it can help screen drug candidates, toxicology and your dose range using these models.
Translational research requires you to get much closer to a human model. Non-human primates (NHPs), specifically cynomolgus monkeys, are considered the most clinically relevant. They have structural and functional similarities to humans, meaning any results are similar to what we would anticipate to see if we were to dose a human. The monkeys experience similar adverse effects and are actually pretty compatible for study with imaging techniques used on humans like MRIs, CTs, etc. The downside is that NHPs are super expensive, many thousands of dollars, and the monkeys don’t live long. They have a lot less cerebrospinal fluid than humans, and they’re a lot harder to dose in the intrathecal space — the space between the spinal cord and vertebrae — because they’re a much smaller animal, so the space is super tight compared to a human’s.
That’s where ex vivo testing comes in, where you’re using tissue that’s already been excised from an animal. That tissue is no longer alive, but it’s fresh enough that you can still inject into it. Our recommendation would be to use pigs because they’re bigger, and that way we get a more representative idea of tissue properties of large mammals and how they’ll hold up against repeated injection. The downside is that pigs are fat, to put it bluntly, so you have to go through a lot more effort to get to the intrathecal space, which means the procedure involved is way more invasive. Plus, pigs are quadrupeds, so their neural axis is built differently than humans. It’s on a horizontal axis, whereas humans are on a vertical axis because they’re bipedal. In that sense, when it comes to studying drug distribution, you may not get as much valuable data out of that, but pigs are still worth looking into since they’re a larger animal model and therefore have slightly more representative tissue properties to that of humans.
Why is translating findings from animal to human models important?
Where this method of drug delivery gets tricky is the volume of the drugs involved. The volumes that (pharmaceutical companies are)/ Lilly is looking into are very large and meant to be given over a longer duration of time, like a period of 10 to 20 minutes. You’ll naturally lose a decent amount of the drug as you inject it, so you have to inject a lot more to maximize the benefits you get. You have to make sure you’re not introducing a level of toxicity that patients won’t be able to tolerate. Hence the importance of translational research. Test it out in animals so you’re not delivering a toxic dose to a human by the time you get to that level of study.
WHY IT MATTERS
What are some of the implications of this research?
The main difficulty in research like this is trying to find a one-size-fits-all solution. That just doesn’t work here. You need to come up with a tiered approach like the one we discuss in this publication. On top of that, you need to find a way to validate that the aspect of research that you’re demonstrating actually holds true in a human setting.
I hope people can use our work as an opportunity to standardize best practices for drug delivery research, not just for central nervous system delivery, but for any other drug delivery system that goes beyond the common injection into the skin or bloodstream like you get at the doctor’s office. The publication cites a good precedent, or at least, supporting evidence of how to do good research.
How might this publication continue to affect your work with ASG?
We’re starting to see a slow increase in areas of work that fall under a space of engineering research. It’s going a little beyond doing work that follows a standard operating procedure. It’s applying observations and plans in a biological context to define a drug delivery system, which is a lot of what ASG does. We help provide solutions that develop drug delivery systems to better human lives.
I’ve published something on behalf of ASG in support of a client; that’s huge! I’m hoping that our team will have more opportunities like this. Whether it’s me speaking about it in tandem with my colleagues at conferences or more people getting opportunities to do a similar type of work, I can see this growing into more opportunities to showcase what we can do and really emphasize the diversity of services we provide.
Read the full article by Suna and her team.
Contact us to learn more about ASG.
Medical illustrations from “A multimodal perspective to overcome translational barriers in cerebrospinal drug delivery” published in Expert Opinion on Drug Delivery (EODD)





